Spirometry Respiratory and COPD
Spirometry test machines measure lung function to assist in the diagnosis and management of respiratory conditions. These devices support clinical assessment for asthma and COPD within primary care settings.
More Information
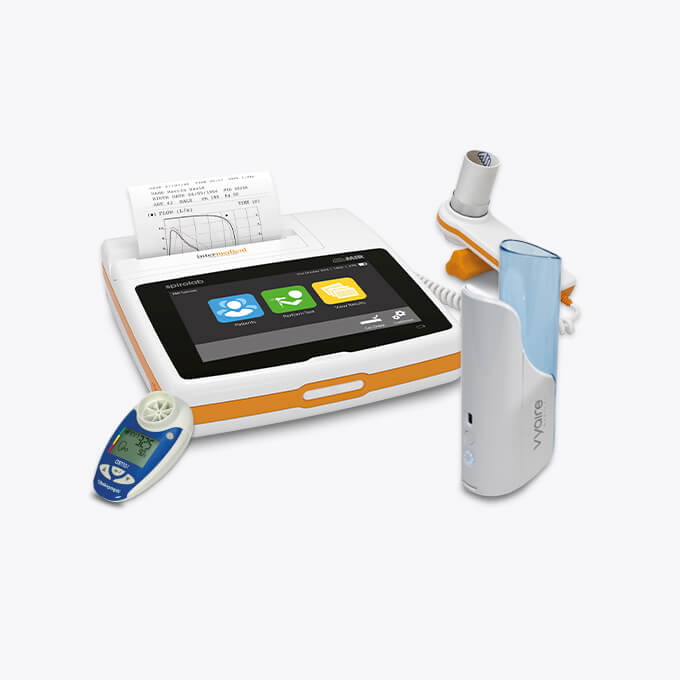
Desktop, handheld & PC Based Spirometers
Discover high-quality spirometry, respiratory, and COPD equipment at Williams Medical. Equip your medical facility with reliable tools for accurate diagnosis and improved respiratory management.
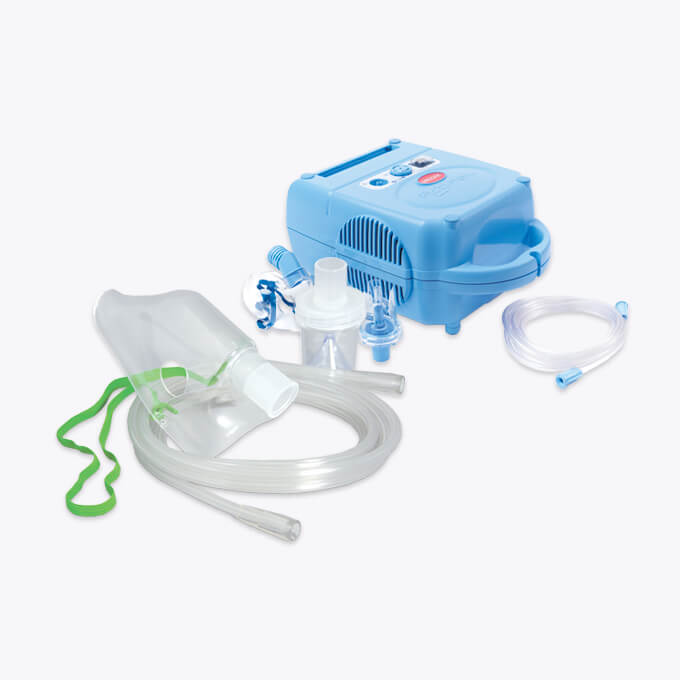
Nebuliser
We stock a range of nebulisers and a seclection of adult and paediatric masks

Peak Flow
EU Standard Peak flow meters suitable for adults and children, allowing patients to monitor their asthma.

Spacers
Improved delivery of Asthma medication with the our range of Asthma spacers
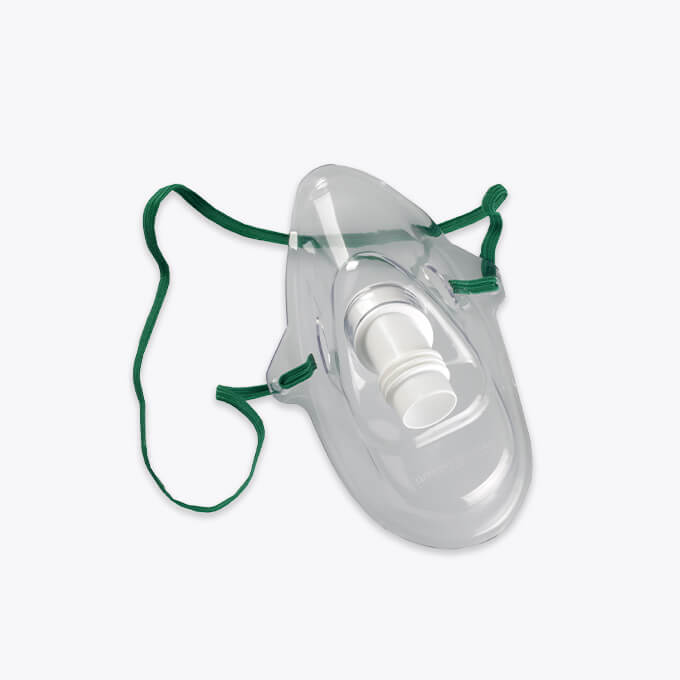
Masks
Browse our range of nebulisers and oxygen masks suitable for adults and children

Accessories
Here you'll find thermal paper, inhaler trainers and a range of other respiratory accessories
Frequently Asked Questions Williams Medical Supplies provides spirometry and respiratory diagnostic equipment for UK primary care professionals. The following FAQs reflect information drawn from recognised clinical standards and regulatory guidance. What's the difference between a handheld and desktop spirometer? Handheld spirometers are battery-powered and portable, designed for use across a range of clinical settings. Desktop spirometers typically feature integrated printers, larger displays, and more advanced software for reviewing results such as FEV1/FVC trends and flow-volume loops. Manufacturers of desktop devices generally describe them as suited to fixed treatment room environments with higher testing volumes. The suitability of either type for a given practice will depend on clinical workflow, available space, and intended use — decisions that are best made in consultation with a clinical lead or equipment supplier. What should I look for when buying a spirometer for my GP surgery? ATS/ERS standards identify several technical requirements that spirometry equipment must meet, including accuracy thresholds for volume and flow measurement. Beyond compliance, clinicians commonly evaluate transducer type (turbine or pneumotachograph), compatibility with clinical systems such as EMIS Web or SystmOne, on-board data storage, and support for automatic ATS/ERS quality grading of test results. Infection control considerations, including compatibility with single-use bacterial/viral filters, are also noted in ARTP guidance as relevant to equipment selection in primary care settings. What standards and regulations apply to spirometers sold in the UK — and does my device need to be UKCA marked? Spirometers sold in the UK must comply with ISO 26782, the international standard governing the accuracy and performance of lung function testing equipment. Devices must also meet the performance criteria set out by the American Thoracic Society and European Respiratory Society (ATS/ERS). Under the UK Medical Devices Regulations 2002 (as amended), devices must carry either CE or UKCA marking to be legally placed on the UK market. The Association for Respiratory Technology and Physiology (ARTP) publishes separate guidance on equipment standards that is widely referenced by NHS commissioners and practice managers. How do I calibrate a spirometer, and how often should calibration be performed? ATS/ERS guidelines recommend that spirometers are calibrated using a 3-litre calibration syringe, with checks performed before each testing session. The syringe delivers a known volume to verify the device is reading within acceptable accuracy thresholds. ARTP guidance also references biological quality control, in which a trained operator with stable lung function performs a reference test periodically to help identify device drift over time. For specific calibration schedules and servicing intervals, manufacturers' instructions should be followed. What infection control measures apply to spirometry testing in UK primary care? ARTP and NHS infection prevention and control (IPC) guidance indicates that a single-use bacterial/viral filter or disposable flow transducer should be used for each patient to prevent cross-contamination of the device's internal pathways. Reusable components should be decontaminated in line with the device manufacturer's instructions and local IPC policy. Practices are directed to their local IPC lead and the relevant manufacturer documentation for specific decontamination procedures applicable to their equipment. What routine maintenance does a spirometer require? Manufacturer instructions for individual devices set out the specific maintenance requirements for that equipment. Generally, ATS/ERS guidance identifies daily calibration checks and flow sensor inspection as part of quality control in spirometry. Software and firmware updates are typically issued by manufacturers to maintain compliance with current ATS/ERS quality grading algorithms. For servicing intervals and component replacement schedules, practices should refer to the manufacturer's documentation or contact their equipment supplier. Do spirometers integrate with EMIS Web or SystmOne? A number of desktop spirometers are designed with PC connectivity to support integration with clinical systems, including EMIS Web and SystmOne. Where available, this functionality allows spirometry results — including FEV1, FVC, and FEV1/FVC ratio — to be transferred directly into patient records. Integration capability varies by device and software version. Practices are advised by system suppliers to confirm compatibility between the spirometer's software and their specific clinical system installation prior to purchase. Does spirometry carried out in my practice count towards QOF — and what equipment requirements apply? Under the NHS Quality and Outcomes Framework (QOF), spirometry is required for confirming diagnoses within the COPD register domain. NHS England and NICE guideline NG115 specify that a post-bronchodilator FEV1/FVC ratio of below 0.70 is used to confirm airflow obstruction consistent with COPD. ATS/ERS quality criteria apply to the acceptability of individual tests. The ARTP Certificate of Competence in Spirometry is widely referenced in NHS commissioning guidance as the recognised qualification for operators performing diagnostic spirometry in primary care. What do FEV1, FVC, and FEV1/FVC ratio mean in spirometry results? FEV1 (Forced Expiratory Volume in 1 second) is the volume of air exhaled in the first second of a maximal forced exhalation and is used as an indicator of airflow obstruction. FVC (Forced Vital Capacity) is the total volume of air exhaled during the full manoeuvre. The FEV1/FVC ratio expresses FEV1 as a proportion of FVC. NICE guideline NG115 references a post-bronchodilator FEV1/FVC ratio below 0.70 as consistent with airflow obstruction in the diagnosis of COPD. Interpretation of spirometry results should be carried out by a trained and competent clinician. What's the difference between a turbine and pneumotachograph spirometer? Turbine spirometers measure airflow by detecting the rotation of a small impeller moved by exhaled breath. Pneumotachograph spirometers measure airflow by detecting the pressure difference across a fine mesh screen. Manufacturers describe differing maintenance and handling requirements for each transducer type. The ARTP and ATS/ERS both note that transducer type can affect calibration requirements and susceptibility to environmental factors such as moisture. For guidance on which transducer type is appropriate for a specific clinical setting, manufacturers' product documentation and clinical leads should be consulted. Why is my spirometer giving inconsistent or low readings — what should I check? ATS/ERS quality control guidance identifies several factors that can contribute to inconsistent spirometry results, including device calibration drift, flow sensor contamination, and patient technique — such as insufficient expiratory effort or early termination of the manoeuvre. Manufacturer troubleshooting documentation for individual devices sets out the steps for diagnosing equipment-related causes. Where calibration checks or visual inspection do not resolve the issue, contacting the device manufacturer or equipment supplier is the appropriate course of action.



